
In vitro Diagnostics (IVD) Market
In vitro Diagnostics (IVD) Market - Global Industry Assessment & Forecast
Segments Covered
By Products & Services Reagents & Kits, Instruments, Services, Data Management Software
By Technology Immunoassay/ Immunochemistry, Clinical Chemistry, Molecular Diagnostics, Hematology, Microbiology, Coagulation & Hemostasis, Urinalysis, Other Technologies
By Application Infectious Diseases, Diabetes, Oncology, Cardiology, Drug Testing/ Pharmacogenomics, Autoimmune Diseases, HIV/AIDS, Nephrology, Other Applications
By End User Hospital Laboratories, Clinical Laboratories, Point-of-care Testing Centers, Academic Institutes, Patients, Other End Users
By Region North America, Europe, Asia Pacific, Latin America, Middle East & Africa
Snapshot
| 2022 | |
| 2023 - 2030 | |
| 2017 - 2021 | |
| USD 112.64 Billion | |
| USD 157.75 Billion | |
| 4.30% | |
| Asia Pacific | |
| North America |
Customization Offered
Cross-segment Market Size and Analysis for Mentioned Segments
Additional Company Profiles (Upto 5 With No Cost)
Additional Countries (Apart From Mentioned Countries)
Country/Region-specific Report
Go To Market Strategy
Region Specific Market Dynamics
Region Level Market Share
Import Export Analysis
Production Analysis
Others Request Customization Speak To Analyst
Premium Insights
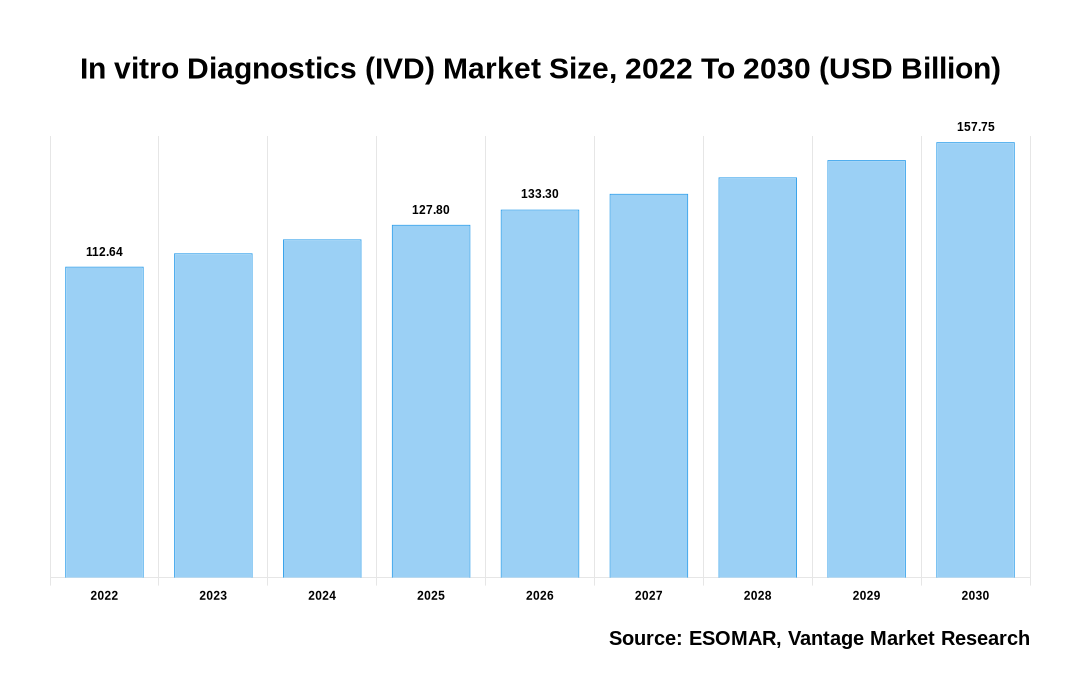
The Global In vitro Diagnostics (IVD) Market is valued at USD 112.64 Billion in 2022 and is projected to reach a value of USD 157.75 Billion by 2030 at a CAGR (Compound Annual Growth Rate) of 4.30% over the forecast period.
The increase in the prevalence of chronic and infectious diseases is a major factor propelling the growth of the In vitro Diagnostics (IVD) market. In today's corporate environment, chronic diseases such as diabetes, cancer, cardiovascular disease, and tuberculosis are on the rise. In addition, there has been a significant rise in the prevalence of infectious diseases such as STDs (sexually transmitted diseases), respiratory infections, and gastrointestinal disorders. The market for In vitro Diagnostics (IVD) is driven by the predicted increase in demand for diagnostic tools as the frequency of certain diseases rises. In addition, recent technological advancements like as rapid test kits have substantially benefited the IVD industry, and this trend is anticipated to continue. In-vitro diagnostics are utilized in many aspects of healthcare. Due to the expensive nature of these tests, however, it is anticipated that patients would seek external options that could restrain the In vitro Diagnostics (IVD) market. In addition, the rate of internal substitution for identifying new diseases, such as SARS-CoV-2, is significant, which stimulates competition amongst rivals. Due to the aging population and a deeper awareness of early diagnosis, the frequency of routine checkups has grown, as the majority of deaths from infections and chronic diseases occur in individuals over 75 years of age.
In vitro Diagnostics (IVD) Market Size, 2022 To 2030 (USD Billion)
AI (GPT) is here !!! Ask questions about In vitro Diagnostics (IVD) Market
Market Segmentation
The In vitro Diagnostics (IVD) market is segmented on the basis of Products & Services, Technology, Application, End User, and Region. The Products & Services market is segmented as Reagents & Kits, Instruments, services, and Data Management Software. Furthermore, based on the Technology, the market is further divided into Immunoassay/ Immunochemistry, Clinical chemistry, Molecular diagnostics, Hematology, Microbiology, Coagulation and Hemostasis, Urinalysis, and Other Technologies. In addition, on the basis of the Applications, the market is segmented as Infectious diseases, Diabetes, Oncology, Cardiology, Drug Testing/ Pharmacogenomics, Autoimmune diseases, HIV/AIDS, Nephrology, and Other applications. Additionally, based on the End User, the market is further bifurcated into Hospital laboratories, Clinical laboratories, Point-of-care testing centers, Academic institutes, Patients, and Other End Users. Likewise, based on the Region, the Global In vitro Diagnostics (IVD) Market is fragmented into North America, Europe, Asia Pacific, Latin America, Middle East & Africa.


Report Coverage & Deliverables
- Real-Time Data Updates:
- Competitor Benchmarking
- Market Trends Heatmap
- Custom Research Queries
- Market Sentiment Analysis
- Demographic and Geographic Insights
Get Access Now
Based on Technology
The molecular diagnostic category dominated the market in 2021
Molecular diagnostics is expected to dominate the market share during the forecast period. The factors influencing the growth of this market include new product introductions and recent regulatory approvals, simple access to a large selection of reagents, and the appearance of cutting-edge technologies like qRT-PCR. In addition, the rise of the coronavirus pandemic in the last few years has accelerated the development of molecular diagnostic methods because numerous manufacturers have introduced IVD solutions for diagnosing coronavirus based on molecular diagnostic methods.
Based on Applications
The infectious diseases category dominated the market in 2021
The infectious disease segment is anticipated to grow at the highest rate during the forecast period. The largest share of this market is primarily attributable to the rise in the prevalence of infectious diseases worldwide, the aging population's increased susceptibility to disease, the emphasis on early disease detection using particular diagnostic tests, government initiatives to improve the accessibility of diagnostic services for infectious diseases, and the emergence of the coronavirus pandemic in the recent times.
Based on Regional Analysis-
North America is projected to grow at the highest CAGR during the forecast period.
During the projected time, North America occupies a sizeable portion of the market. This is attributable to the region's well-integrated healthcare system, patients' increasing awareness of healthcare issues, patients' adoption of innovative therapies, and overall growth in healthcare spending. However, North America is also predicted to rise substantially due to rising healthcare expenditures, a large patient population with chronic diseases requiring IVD testing and rising disposable incomes. The region is anticipated to continue to dominate the market throughout the forecast period. This region's sector is driven by a number of factors, including the rising frequency of chronic diseases, the rapidly expanding senior population, high healthcare expenses, and supporting government funding. In North America, the demand for genetic testing to enable personalized healthcare, such as for diabetes and cancer, is predicted to fuel market growth.
Competitive Landscape
The key players in the In vitro Diagnostics (IVD) market include- Roche Diagnostics (Switzerland), Siemens Healthineers (Germany), Danaher Corporation (US), Abbott (US), Thermo Fisher Scientific (US), Johnson & Johnson (US), Becton Dickinson & Company (US), Bio-Rad Laboratories (US), Sysmex Corporation (Japan), bioMérieux (France), DiaSorin (Italy), Ortho Clinical Diagnostics (US), QIAGEN N.V. (Netherlands) among others.
Recent Developments
· In February 2022, LiquidPlex Dx and FusionPlex Dx cancer testing kits were launched by Invitae to allow efficient management of cancer patients with therapy with timely provision of the required information.
· In January 2022, Roche Diagnostics (Switzerland) launched the Cobas pulse system, a blood glucose management solution with mobile digital health capabilities to improve patient care.
· In September 2021, Abbott Laboratories (US) received US FDA clearance for the Freestyle libre 2 iOS application for use with compatible iPhones
· In March 2021, Siemens Healthineers renewed its long-standing supply, distributorship, sales, and service agreement for hemostasis, including a multi-year extension with Sysmex Corporation (Japan).
Segmentation of In vitro Diagnostics (IVD) Market
| Parameter | Details |
|---|---|
| Segments Covered |
By Products & Services
By Technology
By Application
By End User
By Region
|
| Regions & Countries Covered |
|
| Companies Covered |
|
| Report Coverage | Market growth drivers, restraints, opportunities, Porter’s five forces analysis, PEST analysis, value chain analysis, regulatory landscape, technology landscape, patent analysis, market attractiveness analysis by segments and North America, company market share analysis, and COVID-19 impact analysis |
| Pricing and purchase options | Avail of customized purchase options to meet your exact research needs. Explore purchase options |
 Vantage Market
Research | 15-Dec-2022
Vantage Market
Research | 15-Dec-2022
FAQ
Frequently Asked Question
What is the global demand for In vitro Diagnostics (IVD) in terms of revenue?
-
The global In vitro Diagnostics (IVD) valued at USD 112.64 Billion in 2022 and is expected to reach USD 157.75 Billion in 2030 growing at a CAGR of 4.30%.
Which are the prominent players in the market?
-
The prominent players in the market are Roche Diagnostics (Switzerland), Siemens Healthineers (Germany), Danaher Corporation (US), Abbott (US), Thermo Fisher Scientific (US), Johnson & Johnson (US), Becton Dickinson & Company (US), Bio-Rad Laboratories (US), Sysmex Corporation (Japan), bioMérieux (France), DiaSorin (Italy), Ortho Clinical Diagnostics (US), QIAGEN N.V. (Netherlands).
At what CAGR is the market projected to grow within the forecast period?
-
The market is project to grow at a CAGR of 4.30% between 2023 and 2030.
What are the driving factors fueling the growth of the market.
-
The driving factors of the In vitro Diagnostics (IVD) include
- Rising geriatric population and subsequent growth in the prevalence of chronic and infectious diseases
Which region accounted for the largest share in the market?
-
North America was the leading regional segment of the In vitro Diagnostics (IVD) in 2022.



